- Home
- Industry
- Applications and case studies
- Renewable Energy and Energy Storage
Renewable Energy and Energy Storage
It is clear by now that the supply of fossil energy is limited. Furthermore, returning large quantities of carbon dioxide to the atmosphere increases the amount of energy that it absorbs from the sun with consequences for the balance of our planet’s climate systems. There is a need to develop alternative sources of energy and to ensure that energy, irrespective of its source, is used as efficiently as possible, which then results in that energy having to be stored, delivered and converted into heat, light or motion with minimal losses.
Materials research is at the heart of developments for modern energy systems. The ESRF offers the possibility of studying energy producing or energy storage systems, from photovoltaic panels, to fuel cells or rechargeable batteries or even hydrogen-storage materials. Some of the techniques that can be used are:
- Diffraction and spectroscopy to reveal the composition, atomic structure and crystalline state of materials.
- Imaging of materials to track defects and aggregates.
- In situ studies to follow materials like batteries under operating conditions or to watch the evolution of components.
Company or institute
CNRS and Renault
Challenge
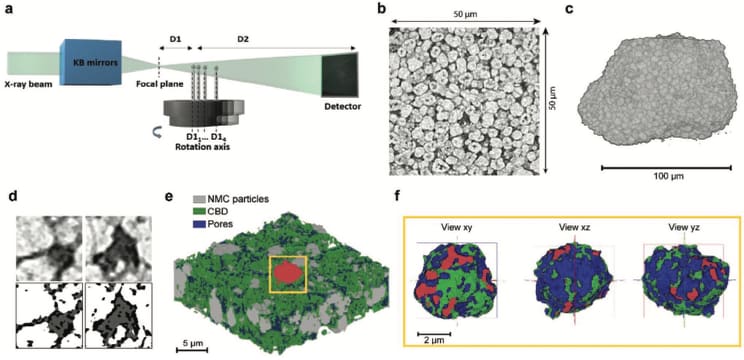
The aim of this research was to visualise and quantify the components that constitute a nickel, manganese and cobalt (NMC) cathode. These components are the NMC material, the carbon binder domains (CBD) and the network of pores that enable the liquid electrolyte to fill the electrode. The particular challenge was to reveal the morphology of the CBD, complicated by its low X-ray attenuation coefficient.
Sample
The samples were 50 micrometre wide sections cut from LiNi0.5Mn0.3Co0.2O2 high energy density cathodes of different compositions containing a mixture of NMC, conductive carbon black and binder (e.g., polyvinylidene fluoride (PVdF)).
Solution
At beamline ID16B, quantitative phase-contrast X‐ray holotomography was used to image the cathode samples at the nano-scale. This 3D method provided a large reconstructed volume, where the CBD could be resolved along with the active materials and the pore space. The spatial relation of the three components was mapped in an ensemble of 500 individual active particles with a spatial resolution of 50 nm in a volume of 50x60x60 µm3.
Benefits
The imaging method provided volumes with the required resolution and contrast to perform complete quantitative analysis of the microstructure of the cathode including the characterisation of the active material, the CBD and the porosity separately and their inter‐connectivity at the particle scale. The results provided an understanding of the negative impact of the CBD excess on electrode performance at high charging/discharging rates. The impact of these results will be in the optimisation of the electrode design to improve the power rate of high energy density electrodes.
This research was also featured in an ESRF news item: Inside the 3D electrode architecture: towards better performing batteries for electric cars.
Reference
3D Quantification of Microstructural Properties of LiNi0.5Mn0.3Co0.2O2 High‐energy density electrodes by X‐ray holographic nano‐tomography, Nguyen, T.‐T., Villanova, J., Su, Z., Tucoulou, R., Fleutot, B., Delobel, B., Delacourt, C., Demortière, A., Adv. Energy Mater., 2003529 (2021); DOI: 10.1002/aenm.202003529.
Company or institute
National Renewable Energy Laboratory, Golden, USA.
Challenge
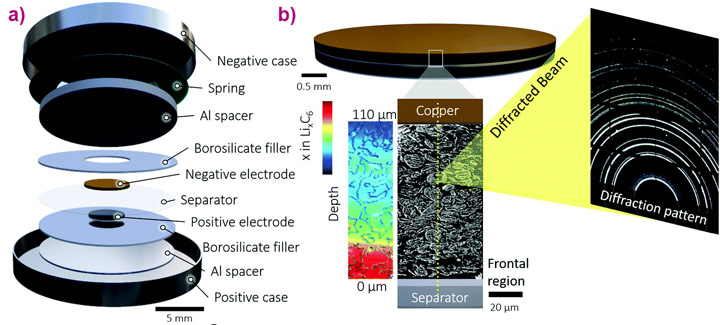
The researchers aimed to better understand the role of lithium plating, which hinders fast charging of lithium-ion batteries. The challenge at beamline ID15A was to visualise the lithiation of a graphite electrode under operando conditions during fast charging and in particular to track the changes throughout the depth of the material, from separator to current collector.
Sample
A standard CR2032 coin-cell format was used to contain the sample, which was a thick (101 μm) graphite electrode.
Solution
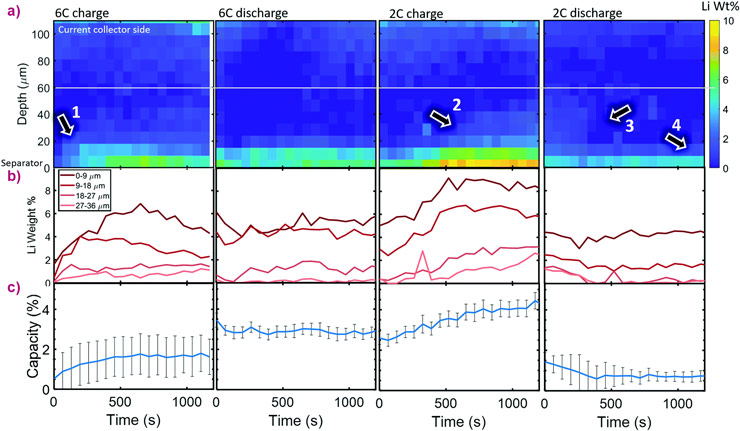
High-speed (100 Hz) synchrotron X-ray diffraction (XRD) was used to investigate lithiation and lithium plating dynamics as a function of depth and time under fast charge and discharge conditions of up to 6C (full charge in 10 minutes). Each complete depth profile was captured in 0.5 seconds, using steps of 3 μm.
Benefits
This study provided the first spatial and temporal description of depth heterogeneities of lithium plating, current densities, and state-of-charge during fast charge and discharge conditions. Significant lithium plating was observed only within the first 20 μm from the separator. Lithium plating changed the behaviour of the underlying graphite, such as causing co-existence of the LiC6 phase and graphite in the fully-discharged state.
Reference
Spatial dynamics of lithiation and lithium plating during high-rate operation of graphite electrodes, D.P. Finegan, A. Quinn, D.S. Wragg, A.M. Colclasure, X. Lu, C. Tan, T.M.M. Heenan, R. Jervis, D.J.L. Brett, S. Das, T. Gao, D.A. Cogswell, M.Z. Bazant, M. Di Michiel, S. Checchia, P.R. Shearing and K. Smith, Energy Environ. Sci., 2020; DOI: 10.1039/d0ee01191f.
This article has also been featured as an ESRF News item.
Company or institute
Imperial College London and Finden
Challenge
To carry out a complete physical and chemical characterisation of an operating ceramic fuel cell, in particular, probing its 3D structure, chemical homogeneity, and mechanical robustness.
Sample
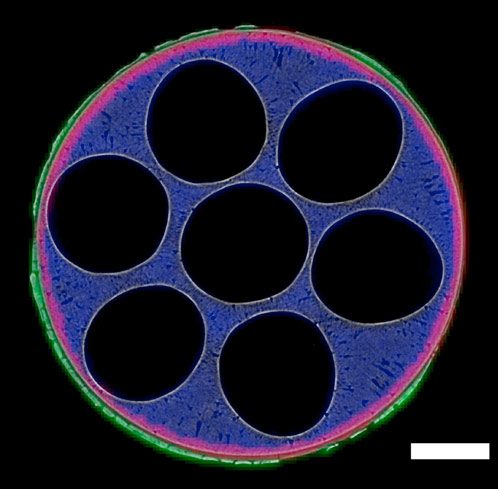
The sample was a miniaturised ceramic solid oxide fuel cell (SOFC) designed for portable applications. Its operating temperature is 800°C.
Solution
A combination of techniques were used together at beamline ID15A. Micro computed tomography (micro-CT) was used to obtain the morphology of the cell, while X-ray diffraction computed tomography (XRD-CT) was used to map the chemical species present within the materials.
Benefits
Micro-CT provided a detailed view of the morphology of the cell, mapping its porous structure and verification that this was unchanged by thermal cycling. In situ XRD-CT provided a confirmation of stability of the materials, by monitoring the crystallographic properties during thermal cycling. From the XRD-CT data, a map of the three electrochemical components of the cell was produced and it was even possible to identify minor phases such as SrO present at < 3 wt.%. Also, the activation of NiO to Ni was observed in the reduction step before cycling.
The combination of techniques provided a unique insight into the functioning of a novel SOFC, demonstrating a very powerful technique to study fuel cells under operando conditions.
Reference
Design of next-generation ceramic fuel cells and real-time characterization with synchrotron X-ray diffraction computed tomography, T. Li, T.M.M. Heenan, M.F. Rabuni, B. Wang, N.M. Farandos, G.H. Kelsall, D. Matras, C. Tan, X. Lu, S.D.M. Jacques, D.J.L. Brett, P.R. Shearing, M. Di Michiel, A.M. Beale, A. Vamvakeros and K. Li, Nature Communications 10, 1497 (2019); doi: 10.1038/s41467-019-09427-z.
Company or institute
University College London
Challenge
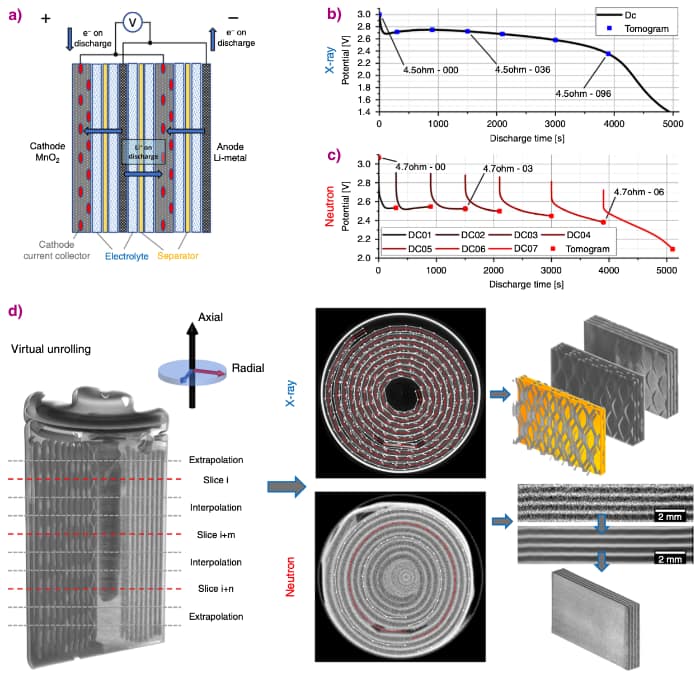
Lithium-ion batteries degrade through damage such as cracks occurring within the electrodes because their sizes change as lithium moves between the electrodes. This study aimed to follow the 3D evolution of a commercial battery during discharge non-destructively, using complementary X-ray and neutron imaging.
Sample
Commercial CR2 Li-ion primary cells were studied (manufacturer Duracell).
Solution
X-ray tomography at beamline ID15A was used to track the battery structure evolution during discharge: 103 tomograms were taken at a rate of one every 40 seconds, with each tomogram lasting 2.8 seconds. Complementary neutron imaging was carried out at the beamline V7 CONRAD-2 of BER II, Helmholtz Centre Berlin.
Benefits
The combination of X-ray and neutron computed tomography provides powerful complementary information. Mechanical degradation processes such as cracking, delamination or unravelling can be studied using X-rays, while Li depletion from the anode or its insertion into the cathode can be inferred by observing the electrode swelling and the loss of image intensity. Whereas neutrons, being highly sensitive to Li, can be used to track light components such as Li metal, Li ions, Li salt as well as the hydrogen in the electrolyte.
Virtual unrolling of the tomograms permitted direct comparison of the images, providing quantitative data such as the expansion of the cathode, and a means to map the distribution of lithium.
Reference
4D imaging of lithium-batteries using correlative neutron and X-ray tomography with a virtual unrolling technique, R.F. Ziesche, T. Arlt, D.P. Finegan, T.M.M. Heenan, A. Tengattini, D. Baum, N. Kardjilov, H. Markötter, I. Manke, W. Kockelmann, D.J.L. Brett & P.R. Shearing, Nature Communications 11, 777 (2020); DOI: 10.1038/s41467-019-13943-3.